Conformational landscape of neurotransmitters
Combining molecular-beam Fourier transform microwave (MB-FTMW) spectroscopy with heating methods, we have characterized the conformational panoramas of several neurotransmitters in the gas phase, including 2-phenylethylamine, p-methoxyphenylethylamine, norephedrine, ephedrine, and pseudoephedrine. However, the investigation of a complete series of neurotransmitters by MB-FTMW spectroscopy is not possible because of the high melting points and associated very low vapor pressures of these compounds. Laser ablation in combination with molecular beams and microwave spectroscopy (LA-MB-FTMW spectroscopy)has proven to be a powerful tool in the investigation of the gas-phase conformational behavior of solid neurotransmitters such as taurine, tryptamine,γ-aminobutyric acid (GABA),serotonin,dopamine, octopamine and synephrine.
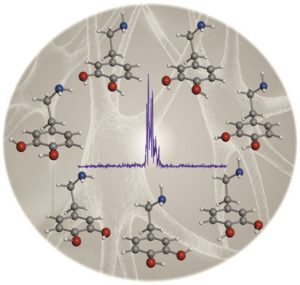
Seven Conformers of Neutral Dopamine Revealed in the Gas Phase,
J. Phys. Chem. Lett., 2013, 4, 486-490
The rotational spectrum of neutral dopamine has been investigated for the first time using a combination of Fourier transform microwave spectroscopy with laser ablation. The parameters extracted from the analysis of the spectrum unequivocally identify the existence of seven conformers of dopamine. 14N nuclear quadrupole coupling interactions have been used to determine the orientation of the amino group probing the existence of stabilizing N–H···π interactions for all observed conformers.
Last contributions
*Conformational Analysis of Octopamine and Synephrine in the Gas Phase,
J. Phys. Chem. A, 2013, 117, 4907-4915
*The microwave spectrum of neurotransmitter serotonin,
Phys. Chem. Chem. Phys. 2012, 14, 13618-13623
*Conformational behaviour of norephedrine, ephedrine and pseudoephedrine,
J. Am. Chem. Soc., 2009, 131, 4320-4326